Alpha Radiation
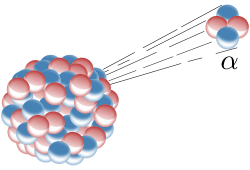
Alpha radiation consists of alpha particles that are made up of two protons and two neutrons. The structure is identical to that of a helium-4 atom without it's electrons. The overall spin of the particle is zero. Alpha particles can be deflected by a magnetic field due to their charge. For the purposes of this article, we will only be looking at alpha particles produced from radioactive alpha decay (those formed from processes such as ternary fission are much higher in energy and therefore penetrating power). The majority of alpha particles have an energy of 3 to 7MeV. The table below summaries the particle's properties:
Ionising ability
| Penetrating Ability
| Stopped by...
| Charge
|
|---|---|---|---|
Very high
| Very low
| A few centimetres of air
| 2e (+2)
|

Alpha decay
Alpha particles are produced in the alpha decay of a heavy atom. Unlike beta and gamma emission, alpha decay can only occur in atoms that have an atomic mass of 106u or over. The decay occurs because of repulsion between two protons - that will make up part of the alpha particle - and the rest of the protons in the atom's nucleus. When the alpha particle is emitted, due to the particle containing two protons, the atom's proton number is reduced by two and therefore, the atom becomes different element - as an atom's element is defined by the number of protons in its nucleus.
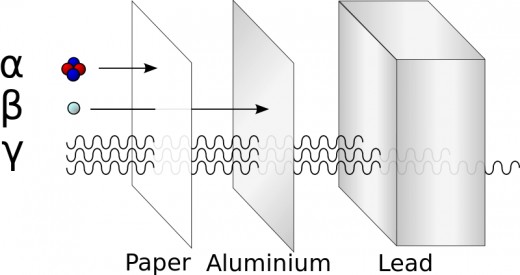
Dangers of alpha radiation
Alpha radiation is generally not considered to be too dangerous. The particles can be stopped by a few centimetres of air, by paper or by skin (once they have lost their energy, they take free electrons from their surroundings and become helium). Therefore, unless ingested or inhaled, alpha radiation poses little threat to humans. However, if the radiation does enter the body, for example, through radon gas, it becomes extremely dangerous as it is the most destructive ionising radiation. The chromosome damage is estimated to be twenty times that of a gamma or beta radiation. Polonium-210 is suggested to be part of the cancerous effects of smoking tobacco as it is often inhaled and ionises cells in the lungs.
Uses
The attribute of alpha radiation that is most commonly exploited is its ability to ionise. It is used in smoke detectors, anti-static devices and small scale generators.
Smoke detectors usually contain a small amount of americium-241, which is an alpha emitter. The americium is placed between two electrical plates. The alpha particles locally ionise the air. A small current is then passed between the plates, being transported between the plates in the ionised air. Smoke will reduce the amount of current that can flow between the plates and if there is sufficient obstruction, the current will drop to such a level that the alarm goes off.
Anti-static devices sometimes contain alpha radiation sources to ionise the air so that the static charge can more easily dissipate.
Alpha emitters are used in some eletrical generators used in objects such as pacemakers. This is because they create a significant amount of heat, which can be converted into electrical energy. They are also usually safe in close proximity to animal cells.
Thank you for reading his article, I hope it has helped you gain a better understanding of alpha radiation. If you have any comments or suggestions, please post them below in the comments section (does not require sign up).
Want to learn about the other types of radiation? Click here
Finally, if you've enjoyed this article, please like, rate or share this article. Want to know more? Check out 'Alpha Radiation'.
Check out my latest article: Essential books on the British Empire